Current Issue
Endoscopic Resection of a Solitary Benign Schwannoma of the Cheek: A Case Report
Sassi Yosra1*, Zheni Alia2, Jed Bouguila1,3,4Sassi Yosra1*, Zheni Alia2, Jed Bouguila1,3,4
1Department of ENT and Maxillofacial Surgery, La Rabta Academic Hospital, Tunis-Tunisia
2Department of Anatomopathology, La Rabta Academic Hospital, Tunis-Tunisia
3Tunis Elmanar University, Tunis-Tunisia
4Laboratory of Oral Health and Maxillofacial Rehabilitation (LR12ES11), Tunisia
*Corresponding author: Dr. Yosra Sassi, MD, Resident, Department of ENT, Maxillo-facial and Aesthetic Surgery. La Rabta Hospital, 1007 Tunis, Tunisia, Phone: Tel: (00216)99625412, E-mail: [email protected]
Citation: Yossra S, et al. (2026). Endoscopic Resection of a Solitary Benign Schwannoma of the Cheek: A Case Report. Cases. 5(1):28.
Received Date: February 22, 2026
Publication Date: April 23, 2026
ABSTRACT
Background: Tumors of the jugal (buccal) region are rare, and their origin is variable. Schwannomas represent a particularly uncommon entity in this location. The standard treatment remains surgical excision, with the approach determined by the location and nature of the lesion. The endoscopic approach appears as a promising minimally invasive alternative, allowing for complete resection while preserving aesthetics. Case Summary: We report the case of a 57-year-old patient presenting with a jugal mass evolving over 10 years, diagnosed as an intramasseteric schwannoma by ultrasound and MRI and confirmed by histopathology with S-100 immunostaining. The patient was treated by endoscopic excision under general anesthesia via two 7 mm incisions, achieving complete tumor removal. The 6-month follow-up showed excellent functional and cosmetic outcomes, with no complications or recurrence. Discussion: Intramasseteric schwannomas are uncommon, benign tumors that develop within the nerve sheath and can be challenging to diagnose and treat due to their deep location. While traditional open surgery provides good access, it can affect facial aesthetics. In this case, using an endoscopic approach allowed for precise tumor removal with minimal impact on surrounding tissues, reducing complications and improving cosmetic results. This minimally invasive technique offers a promising alternative for treating certain buccal tumors, ensuring effective removal while preserving the natural appearance of the face.
Keywords: Buccal Tumor, Intramasseteric Schwannoma, Endoscopic Excision
INTRODUCTION
Tumors in the jugal region, although rare, pose diagnostic and therapeutic challenges due to their proximity to important anatomical structures of the face. Among these tumors, schwannomas—benign tumors arising from Schwann cells—represent a particular entity. Although they can manifest in various regions of the body, their most frequently reported location is the head and neck [1], with only 1% of all head and neck schwannoma cases observed within the oral cavity [2].
The most common site in the head and neck region is the lateral neck, but other locations, particularly the tongue, the submandibular space, and especially the masseter muscle, are rare [3]. In the jugal region, these tumors are rare and can be confused with other subcutaneous lesions such as lipomas, salivary tumors, or cysts [4,5].
We present the case of a jugal schwannoma treated by endoscopic excision and provide a review of the relevant literature on this technique and its application in the masseteric region [6-9].
CASE REPORT
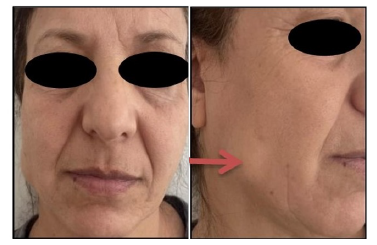
A 57-year-old patient with no notable medical or surgical history presented with a right jugal mass. The patient reported a swelling evolving for about 10 years, initially painless with slow growth, which has become painful in recent months. No history of trauma, dental pain, or salivary colic, nor any other associated signs, was reported. (Figure 1a,1b)
Figure1a: Pre-operative frontal view of the patient
Figure 1b: Pre-operative lateral view of the patient
The clinical examination revealed a low jugal swelling in the area of the horizontal branch of the mandible, measuring approximately 3 cm, firm, mobile, and tender to palpation, without inflammatory signs. The jugal mucosa and the internal buccal wall were intact, with no neurological impairment.
The soft tissue ultrasound showed an oval mass, well-defined, with a hypoechoic and heterogeneous echostructure, measuring approximately 27×23 mm, in direct contact with the bone, without signs of bone erosion.
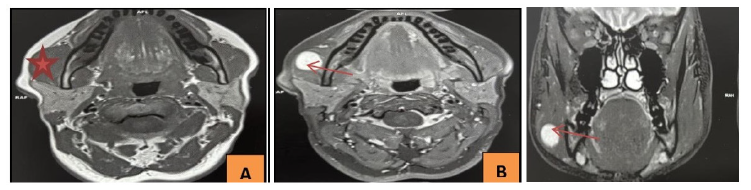
The cervico-facial MRI, performed without contrast injection in T1 and T2 sequences and reviewed by an experienced radiologist, revealed an intra-masseteric process with clear contours (15×18×13 mm) showing a hypointense signal in T1 and a hyperintense signal in T2, compatible with a benign lesion (Figure 2a, 2b, 2c).
Figure 2a: Axial slice in T1: A well-defined intramasseteric mass with hypointense signal on T1
Figure 2b: Axial slice in T2: A well-defined intramasseteric mass with hyperintense signal on T2
Figure 2c: Coronal T2 slice : an intramasseteric mass with hyperintense signal on T2
After a multidisciplinary consultation and the patient's informed consent, an endoscopic excision was performed under general anesthesia. Two discreet incisions of 7 mm each were made: one hidden in a wrinkle in front of the tragus and the other at the root of the helix. The use of a 30° rigid endoscope and specialized endoscopic instruments (dissectors, monopolar cautery) allowed for meticulous dissection under magnified visualization, minimizing tissue trauma and preserving adjacent structures including branches of the facial nerve. The complete resection of the lesion was confirmed intraoperatively. The patient received close postoperative follow-up and was discharged on postoperative day 1 under analgesic treatment. The macroscopic examination revealed a well-defined, non-encapsulated, homogeneous, and whitish nodule measuring 1.2 cm.
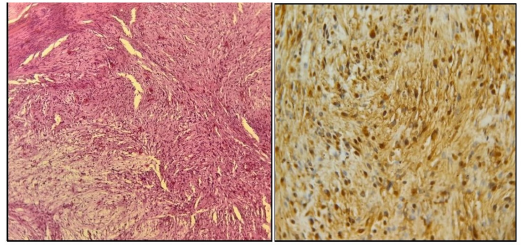
Histological examination, performed by an experienced pathologist, showed a benign tumor proliferation alternating between dense cellular zones and loose myxoid zones, with spindle cells arranged in palisades and hyperchromatic nuclei (Figure 3a).
Immunohistochemistry revealed strong positivity for S-100 protein, confirming the neural origin and the diagnosis of schwannoma (Figure 3b).
Figure 3a: Hematoxylin Eosin x 20: Tumor proliferation composed of spindle cells alternating between dense and loose areas
Figure 3b: Immunohistochemistry x 40: Tumor cells intensely and diffusely express S100 protein
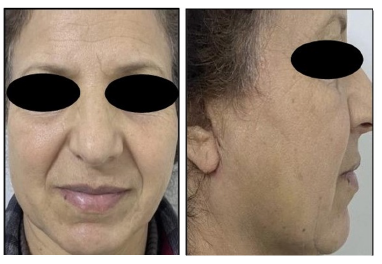
The immediate postoperative follow-up was marked by a rapid recovery and the absence of major complications. The patient noted a significant improvement in pain symptoms from the very first day post-operation. The follow-up assessment at exactly 6 months revealed no functional complaints or complications, no sensory or motor deficits in masseter muscle function, and an excellent cosmetic result (Figure 4a, 4b).
DISCUSSION
Jugal schwannomas, although rare, pose a particular challenge due to their location and the necessity to preserve the functional and aesthetic structures of the face. They can appear on any nerve containing Schwann cells, except for the optic and olfactory nerves, which lack them [10]. The clinical presentation of these tumors is often insidious,
beginning with an asymptomatic phase followed by late symptoms related to the progressive compression of surrounding structures [1].
MRI is the gold standard for characterizing these lesions. T1 and T2 weighted sequences allow for the identification of the typical signal characteristics of schwannomas (hypointensity on T1 and hyperintensity on T2) [4,9]. The clinical signs, associated with a well-defined capsule, suggest a benign lesion. However, in atypical situations, additional tests such as CT scans or percutaneous biopsy may be required to rule out malignancy, especially since MRI does not reliably differentiate a schwannoma from a neurofibroma [11]. Differentiation from neurofibroma is crucial, as the latter may be associated with neurofibromatosis [7,8].
In histopathological analysis, the differentiation between Antoni A and Antoni B areas is an essential diagnostic criterion, supported by strong positivity for S-100 protein, which confirms the neural origin of schwannomas [12,13].
From a future research perspective, molecular alterations such as mutations or loss of expression of the NF2 gene, which have been identified in certain sporadic schwannoma forms, may pave the way for a better biological understanding of these tumors and the development of targeted therapies for recurrent or complex cases. Such molecular profiling was not performed in this case but warrants consideration in future investigations [14].
Although conventional open surgery allows for optimal exposure of the lesion, it is frequently accompanied by notable cosmetic scars and high postoperative morbidity, endoscopic excision, thanks to its minimally invasive approach, offers numerous advantages:
- Precision of dissection: Enhanced visualization of the surgical field allows for meticulous dissection and optimal respect for adjacent anatomical structures.
- Reduction of tissue trauma: The technique minimizes skin incisions and the risk of collateral damage, reducing postoperative morbidity.
- Optimization of aesthetic results: The absence of significant scars is a major advantage, particularly in the facial region.
- Accelerated recovery: A reduction in hospitalization time and a quicker return to daily activities have been observed.
However, this approach requires a rigorous patient selection, particularly for well-circumscribed benign tumors. Despite its benefits, the endoscopic approach has certain limitations. The learning curve is significant, and access to certain deep locations can prove to be complex.
Moreover, the risk of recurrence in the case of incomplete resection remains a major concern. The integration of intraoperative imaging techniques, such as ultrasound or fluorescence, as well as the rise of robot-assisted surgery, represents areas for improvement likely to overcome these limitations [15].
As a future direction, technological advancements such as intraoperative imaging (ultrasound or fluorescence) and robot-assisted surgery may further improve surgical precision, optimize tumor delineation, and expand indications for this technique. However, these approaches remain to be validated in prospective studies.
CONCLUSION
Endoscopic excision of jugal schwannomas represents an innovative and effective therapeutic option, combining complete resection with optimal preservation of aesthetic and functional structures.
In carefully selected patients with well-circumscribed intramasseteric schwannomas, endoscopic excision can achieve complete resection with good functional and aesthetic outcomes, while reducing postoperative morbidity and recovery time compared to open surgery. However, a rigorous selection of patients and technical mastery remain essential to ensure the safety and effectiveness of the procedure. Larger case series will be needed to further validate this approach.
ETHICS STATEMENT AND PATIENT CONSENT
Written informed consent was obtained from the patient for treatment, surgical intervention, pathological analysis, and publication of this case report, including all clinical photographs. This study was conducted in accordance with the Declaration of Helsinki.
REFERENCES
- Bayindir T, Kalcioglu MT, Cicek MT, Karadag N, Karaman A. (2013). Schwannoma with an Uncommon Upper Lip Location and Literature Review. Case Reports in Otolaryngology. 2013:363049.
- Yang SW, Lin CY. (2003). Schwannoma of the upper lip: case report and literature review. American Journal of Otolaryngology. 24(5):351?354.
- Wang HK, Gong YL, Wang RX, Zheng XT, Huang SY, Zhang DS. (2016). A rare occurrence of intramasseteric schwannoma – case report and literature review. Revue de stomatologie, de chirurgie maxillo-faciale et de chirurgie orale. 117(3):170?172.
- Lambade PN, Palve D, Lambade D. (2015). Schwannoma of the Cheek: Clinical Case and Literature Review. Journal of Maxillofacial and Oral Surgery. 14(2):327?331.
- Colreavy MP, Lacy PD, Hughes J, Bouchier-Hayes D, Brennan P, O’Dwyer AJ, et al. (2000). Head and neck schwannomas – a 10 year review. The Journal of Laryngology and Otology. 114(2):119?124.
- Moreno-García C, Pons-García MA, González-García R, Monje-Gil F. (2014). Schwannoma of Tongue. Journal Of Maxillofacial and Oral Surgery. 13(2):217?221.
- Wright BA, Jackson D. (1980). Neural tumors of the oral cavity. Oral Surgery, Oral Medicine, and Oral Pathology. 49(6):509?522.
- Williams HK, Cannell H, Silvester K, Williams DM. (1993). Neurilemmoma of the head and neck. The British Journal of Oral & Maxillofacial Surgery. 31(1):32-35.
- Pilavaki M, Chourmouzi D, Kiziridou A, Skordalaki A, Zarampoukas T, Drevelengas A. (2004). Imaging of peripheral nerve sheath tumors with pathologic correlation. European Journal of Radiology. 52(3):229?239.
- Russell DS, Rubinstein LJ, editors. (1989). Tumours of the Peripheral Nervous System. In: Pathology of Tumours of the Nervous System. 5th ed. Baltimore, MD: Williams & Wilkins.
- Binder DK, Smith JS, Barbaro NM. (2004). Primary brachial plexus tumors: imaging, surgical, and pathological findings in 25 patients. Neurosurg Focus.16(5):1?6.
- Stefansson K, Wollmann R, Jerkovic M. (1982). S-100 Protein in Soft- Tissue Tumors Derived From. 106(2): 261-268.
- Chrysomali E, Papanicolaou SI, Dekker NP, Regezi JA. (1997). Benign neural tumors of the oral cavity. Oral Surgery, Oral Medicine, Oral Pathology, Oral Radiology, and Endodontics. 84(4):381?390.
- Neff BA, Welling DB, Akhmametyeva E, Chang LS. (2006). The Molecular Biology of Vestibular Schwannomas: Dissecting the Pathogenic Process at the Molecular Level: Otology & Neurotology. 27(2):197?208.
- Scintu PF, Nord B. (2020). État de l’art de la chirurgie robotique. IRBM News. 41(6):100278.
 Abstract
Abstract  PDF
PDF